Imaging the Sleep Deprived Brain: A Brief Review
Article information
Introduction
Functional magnetic resonance imaging (fMRI) is a highly versatile tool used to study neurobehavioral alterations associated with sleep deprivation (SD). Task-related fMRI is the most widely used technique and an impressive list of cognitive domains has been evaluated using this technique (Table 1).1-40 fMRI measures relative change in blood oxygenation level dependant (BOLD) signal in capillaries and venules adjacent to neuronal clusters whose firing rate and consequently, synaptic potentials are modulated by task performance. An increase in MR signal occurs as a result of a relatively disproportionate elevation in blood flow relative to oxygen consumption in response to sensory stimulation and/or task performance. In addition to task-related activation, the evaluation of task-related deactivation where signal changes fall below baseline levels can be evaluated.1,18,25,30,41
Blood oxygenation level dependant imaging measures relative changes in blood flow, but does not ascertain absolute blood flow. Quantification of blood flow may occasionally be useful, for example, to study time-on-task effects,42 and other phenomena whose observation requires signal stability over several minutes as opposed to several seconds. Such measurements can be obtained using a variety of arterial spin labeling (ASL) techniques that have different levels of precision.43,44 A disadvantage of ASL is its inferior signal to noise ratio relative to BOLD imaging. Additionally, the requirement for block sampling also makes it impossible to perform event-related designs that are important in separating out trials where the subject may have been asleep.8
The evaluation of functional connectivity, conducted by assessing signal covariation in pairs of regions, or by determining the extent to which signal in a ‘target’ region interacts with that of a ‘seed’ region according to state/task context provides additional characterization of altered physiology. The latter method, known as psychophysiological Interaction-PPI has been applied in studies evaluating selective attention,12,45,46 the processing of emotional pictures as well as executive function/working memory.29,35
In addition to fMRI studies designed to evaluate signal changes in response to task performance, it may be informative to evaluate ‘resting-state’ activity or intrinsic functional connectivity.47,48 This refers to the identification of regions showing synchronous low frequency oscillations (0.1-0.01 Hz) in BOLD signal that are not time locked to task performance or sensory stimulation. Studies of this type in sleeping individuals have shown changes in connectivity within the default mode network (DMN) alluded to earlier.49,50 The first study evaluating resting state networks in the setting of SD found selective reductions in DMN functional connectivity and reduced anti-correlation with low frequency oscillations in the ‘task-positive’ network (Fig. 1).18 Analyses of resting state data hold promise of being informative of alterations in brain function without requiring motivated performance on the part of a participant.51 Related to functional connectivity, MRI in the form of diffusion tensor imaging (DTI) can be used to evaluate white matter connectivity. Strangely, only one study to date has used DTI to study sleep deprived individuals.52

Main effect of state on default mode network (DMN) functional connectivity. Schematic showing a significant main effect of state on DMN functional connectivity between 3 node pairs using a seed based analysis; left inferior parietal lobe (LIPL) and dorsal medial prefrontal cortex (dMPFC), LIPL and ventromedial prefrontal cortex (vMPFC), and LIPL and posterior cingulate cortex (PCC; Bonferroni corrected p<0.05/21=0.002). Adapted from De Havas JA, Parimal S, Soon CS, Chee MW. Neuroimage 2012;59:1745- 1751.18 LLTC: left lateral temporal cortex, RLTC: right lateral temporal cortex, RIPL: right inferior parietal lobe.
Combining electroencephalography (EEG) and fMRI in SD or sleep related studies is primarily motivated by the need to monitor sleep stage although the high temporal resolution of EEG is also well suited to study transient phenomena like lapsing.53 The neural correlates of spindles and slow waves have been studied using this technique.54-56 In addition, fMRI guided repetitive transcranial magnetic stimulation has been used to evaluate the therapeutic potential of transcranial magnetic stimulation in alleviating SD.28
Effects of Task Difficulty, Cognitive Domain
Tested and Inter-Individual Differences
The results of behavioral studies suggested that cognitive domain engaged and task difficulty might affect neural response to SD.21 This motivated subsequent studies probing working memory to examine the effect of manipulating task difficulty and/or item load.1,2,57 Although the specific areas most affected by SD differ somewhat across these studies, a common set of findings concerns the decrease in higher visual cortex and fronto-parietal activation when performance declined during SD. In contrast, activation is relatively preserved when performance is maintained. The notion that increased task difficulty might elicit ‘compensatory’ frontal activation was then independently demonstrated in a study evaluating logical reasoning.23
As experience in fMRI cumulated with studies that recruited at least 20 subjects, it became evident that inter-individual differences in response to SD could be identified reliably using imaging. A few studies suggested that a single imaging study conducted in the rested state might predict vulnerability to performance decline in the tested domain whereas later studies found that a shift in activation across state may be more reliable as a marker of inter-individual differences in performance when sleep deprived.4,9,11,24,26
A wealth of behavioral data collected on behavior in sleep deprived persons suggests the loss of vigilance or sustained attention to be the most prominent deficit encountered in SD.14 The failure of attention and the possible maladaptive consequences of endogenous efforts to sustain wakefulness in the face of mounting sleep pressure has been a central idea. A cogent illustration of the vital role played by attention in supporting cognitive performance comes from an experiment originally designed to evaluate visual working memory capacity in SD.5 Participants in this study were briefly shown an array of colored squares and seconds later had to verify if the test item was of a color shown in the array. The number of differently colored squares shown varied from 1 to 8 squares. The key finding was that during SD, even when memory capacity and perceptual load were not taxed, task-related activation in brain regions mediating top-down control of attention and visual perception in extrastriate visual cortex was significantly diminished (Fig. 2).
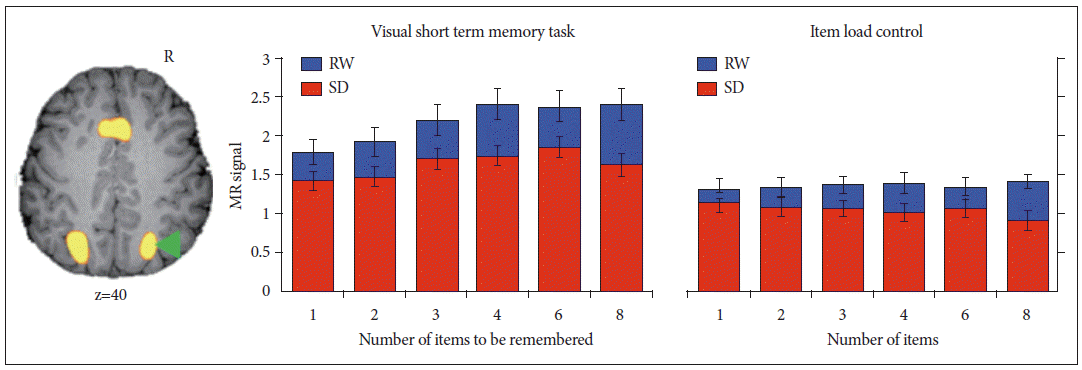
Deficits in attention underlie functional imaging changes when visual short-term memory is tested in the sleep-deprived state. Taskrelated activation of the intraparietal sulcus increases with memory load--but not visual item load when memory is not engaged--in both rested wakefulness (RW) and sleep deprivation (SD). In the sleep-deprived state, even when visual short-term memory is not taxed (e.g., low test item load or no load on memory), decrements in activation are observed. This implicates a fundamental deficit in attention rather than one of storage capacity or memory. Adapted from Chee MW, Chuah YM. Proc Natl Acad Sci U S A 2007;104:9487-9492.5
This indicated that a more global curtailment of processing resources rather than storage limitation was problematic. As both working memory and attention are intertwined processes and engage similar cortical and subcortical areas,58,59 the parametric variation of visual memory and visual item load across multiple levels in two parallel sets of scans were instrumental in proving the point.
Other studies probing the effects of SD on selective attention used visual tracking or visual picture selection paradigms.12,37,46 They arrived at convergent observations regarding the attenuation of top-down control of attention together with diminished engagement of the visual extrastriate cortex. Together with deficits in responding to stimuli, attenuated activation of fronto-parietal and visual extrastriate cortex also occurs in the preparatory period prior to stimulus appearance.15
In comparison to its effects on cortical activation the effects of SD on subcortical activation are more complex. For example, the thalamus, which plays an important role in mediating arousal and attention, shows decreased glucose metabolism during SD if measurement is averaged over several minutes.60 However, for individual events, activation can either be higher or lower than during rested wakefulness depending on whether the sleep deprived volunteer is performing adequately or lapsing.8,37
Evaluating Countermeasures
A majority of persons show decline in cognitive performance after 24 h of SD. fMRI has been used to track the efficacy of countermeasures against, for example, to evaluate the effect of modafinil,39 and donepezil.9,10 In these studies, the effect of drug in the well-rested state was negligible but was evident in the sleep-deprived state in some volunteers. In the donepezil trials, improvement was positively correlated with the magnitude of performance decline when undergoing SD on placebo in both short-term memory and episodic memory experiments in the same subjects. The smaller trial involving modafanil evidenced increases in both cortical and subcortical activation in volunteers sleep deprived on modafanil.
Repetitive transcranial magnetic stimulation has been reported to improve verbal working memory in sleep deprived volunteers when applied to the right ‘upper middle occipital’ region that was part of the network associated with SD induced performance impairment.28 Improvement correlated with the extent to which performance declined during SD in sham stimulations. In contrast no benefit was observed when stimulating the midline parietal region, part of a network of areas showing reduced task-related activation in SD, or when the lower left middle occipital gyrus which was not part this network.
Affective Aspects of Behavior-Emotional Processing and Decision-Making
Most of the tasks used in experimental studies engage a fr-onto-parietal network involved in the control of attention and do not evaluate affective changes that constitute an important aspect of behavioral alteration in SD. Brain structures involved in affective processing include the amygdala, striatum, ventromedial prefrontal cortex and the insula. These structures can be expected to show altered activation if a state-related change in affect is present.
In one study, where participants graded the emotionality of faces, SD enhanced amygdala responses to negative pictures.29 Accompanying this change was reduced functional connectivity between the medial prefrontal cortex and amygdala (Fig. 3). In a second study,13 volunteers remembered neutral faces over a brief interval during which distracter pictures were presented. The distractors could be neutral, negative emotional or noise patterns. Amygdala activation during the maintenance period was higher for emotional distracters than neutral distracters in both states although maintenance activity was overall, lower in the SD state for both conditions (Fig. 3). Face memoranda were less well maintained in working memory to the extent that SD reduced functional connectivity between ventromedial and dorsolateral frontal areas and the amygdala.13 Responses to emotional stimuli during SD can thus be perturbed by disrupting the transmission of top-down control signals from regions involved in valuation and/or cognitive control. In one experiment, negative stimuli were better recognized after SD than neutral or positive stimuli. The recollection of negative and positive stimuli was correlated with greater functional connectivity between medial frontal regions and the hippocampus when subjects had a normal night of sleep compared to when they were sleep-deprived.61
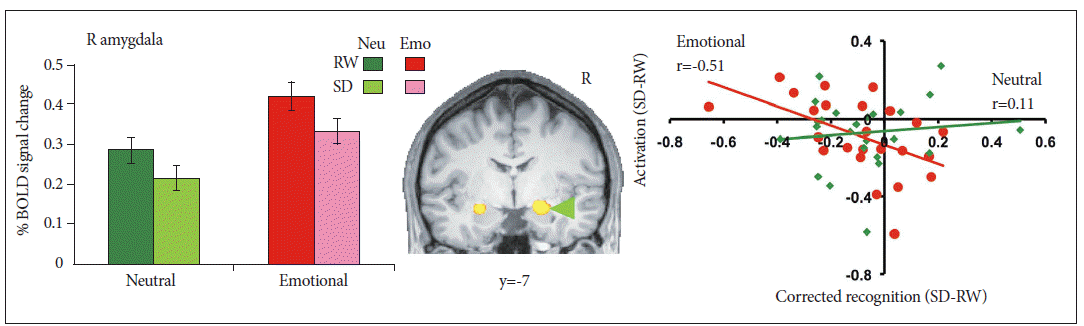
Right amygdala activity was elevated in response to emotional distracters relative to neutral distracters at rested wakefulness (RW). A state (rested wakefulness, sleep deprivation) by Condition (Neutral, Emotional) repeated-measures ANOVA conducted on averaged activation within this region of interest indicated marginal decreases in amygdala activation following sleep deprivation. Critically, state-related change in amygdala activation correlated with the corresponding alteration of emotional distractibility while no parallel effect was present for neutral distracters. Adapted from Chuah LY, Dolcos F, Chen AK, Zheng H, Parimal S, Chee MW. Sleep 2010;33:1305-1313.13 BOLD: blood oxygenation level dependant.
In another experiment, the bias towards processing emotional stimuli over neutral ones was extended to positive stimuli. SD also caused volunteers to increase the number of neutral pictures that were rated positively.32 This finding was related to enhanced hippocampal to medial frontal and hippocampal to orbitofrontal connectivity in the SD state.
Opportunities for Research
A critical gap in our current knowledge of the mechanisms underpinning altered behavior in SD lies in the piecemeal use of functional imaging in this state whereby a single cognitive domain is tested at a time. Behavioral studies show that SD may affect performance unevenly across different cognitive domains.62,63 It remains a open challenge to execute withinsubject studies involving multiple, sensitive, short-duration, tasks that probe dissociable cognitive domains.64
While total SD is convenient to study in a laboratory setting, most persons encounter the effects of sleep loss through chronic sleep restriction and sleep fragmentation.65 Partial SD studies where sleep is restricted to between 3-6 hours of time in bed per night suggest that the relationship between hours of sleep and cognition is not linear.66,67 The rate of build up of slow wave activity during sustained wakefulness and its dissipation following recovery sleep also does not correspond well with behavioral performance alteration suggesting that different measures provide independent information regarding changes occurring in SD. It remains of interest to study how functional imaging changes evolve during partial SD-such studies can provide new understanding of compensatory brain network dynamics during the process of chronic partial sleep restriction.66
Imaging genomics seeks to identify genes that influence brain, cognition and risk for disease. For example, the candidate gene approach was applied to study the effect of Per 35/5 on n-back task performance in subjects undergoing total SD. Widespread relative reductions in task related activation were found in frontal, parietal and occipital regions in this vulner-able group when they were sleep deprived.35 The intriguing behavioral results were not replicated when a different set of sleep restricted (not total SD) subjects were studied leaving room for future studies to clarify.68
Sleep deprivation provides a unique opportunity to perturb brain networks of healthy individuals in a manner that degrades performance in a reversible manner. Several similarities between the behavioral deficits in SD and cognitive aging have been highlighted.69,70 There are also functional imaging parallels that remain to be exploited in future studies.1,71 The attraction of this approach is that it may allow conspecific evaluation of cognitive enhancers in a relatively lower risk setting, keeping in mind that the target group of older adults may be less resilient to adverse effects should these occur.
Acknowledgments
This work was supported by grants awarded to M.C. from the Defence Science and Technology Agency Singapore (POD0713897) and the National Research Foundation Singapore (STaR Award). Substantial portions of this review were derived from Ch. 16 of ‘Neuroimaging of Sleep and Sleep Disorders’ edited by Nofzinger, Maquet and Thorpy; Cambridge University Press 2013.
