Non-rapid eye movement (NREM) parasomnia is defined as recurrent episodes of incomplete awakenings from NREM sleep with inappropriate or absent responsiveness to intervening efforts by others during episodes, and partial or complete amnesia for them. Sleepwalking (SW) is one of NREM parasomnias, typically associated with ambulation and other complex behaviors out of bed [1].
It is considered as a disorder of arousal or a wake-sleep transition disorder [1]. Incomplete arousal, triggered by certain stimuli, from NREM sleep, especially N3 sleep, induce sleep-wake dissociation status with active motor phenomena despite impaired awareness and following amnesia of the events.
SW commonly occurs in childhood. The prevalence of SW increases from 3% at toddler age to 11% at age of 7–8, and peaks at 10 years old (13%)[2]. Children normally grow out of NREM parasomnia during adolescence [2]. The prevalence of adult SW is 2–4%[3,4].NREM parasomnia in adult could occur with triggers including combined sleep disorders, alcohol abuse or medication [3,4]. We report a 25-year-old male patient with severe obstructive sleep apnea (OSA) and a history of spontaneous remission of SW in childhood, who developed recurrent SW and dangerous complex motor behaviors during sleep after severe sleep deprivation.
Case Report
A 26-year-old male patient was presented to the sleep clinic with recurrent episodes of SW and behaviors during sleep, which he could not recall the details. The patient was a first-year resident of the department of cardiothoracic surgery at the University Hospital.
He reports a history of SW from the age of 6 or 7 to his high school years. The trigger was stressful events or being in an unfamiliar environment. He remembered one episode of waking up finding himself walking on the stairs when he was a high school student. No family history of SW or night terror was reported.
No more SW happened since then until recent onset of SW during his internship training. Someone found him yelling and sleepwalking out of dorm 20–30 minutes after he went to bed, and the witness woke him up. The similar events happened more often during the first-year of residency. He sometimes found himself sleeping somewhere different from the original location he started sleeping. One of those episodes was that he woke up at the resident’s office at 2–3 am although he went to bed at 1 am in the dorm. The other episode was that he started sleeping in the dorm on the second floor of the hospital and woke up standing barefoot in the ER on the first floor where was more than 100 meters away from the dorm. Similar episodes occurred almost once every three days.
The most alarming event which finally led him to the sleep clinic was sleepwalking with a smoking during sleep without awareness, resulting in a small fire. After four days of sleep deprivation (three days of on-call nights with almost no sleep and 3 hours of sleep on the following day) he woke up in the middle of the night at the resident’s office, not in the dorm where he first began to sleep. There was a small fire that seemed to be caused by his smoking, which he could not recall. He had a long-standing history of heavy snoring, apnea and bruxism. He had experienced sleep paralysis but denied the presence of cataplexy and hypnagogic hallucination. Epworth sleepiness scale was 20.
The patient’s clinical history was compatible with sleepwalking among NREM parasomnia as he presented recurrent episodes of ambulation without awareness, and with amnesia of episodes. Due to relatively long and variable duration of episodes and no stereotypy associated with behaviors, seizure was clinically ruled out. He denied any use of substance or medication.
Physical examination revealed Mallampati classification grade I, high and narrow hard palate, mild maxillary deficiency and grade III tonsillar hypertrophy. Body mass index was 25.9 kg/m2.
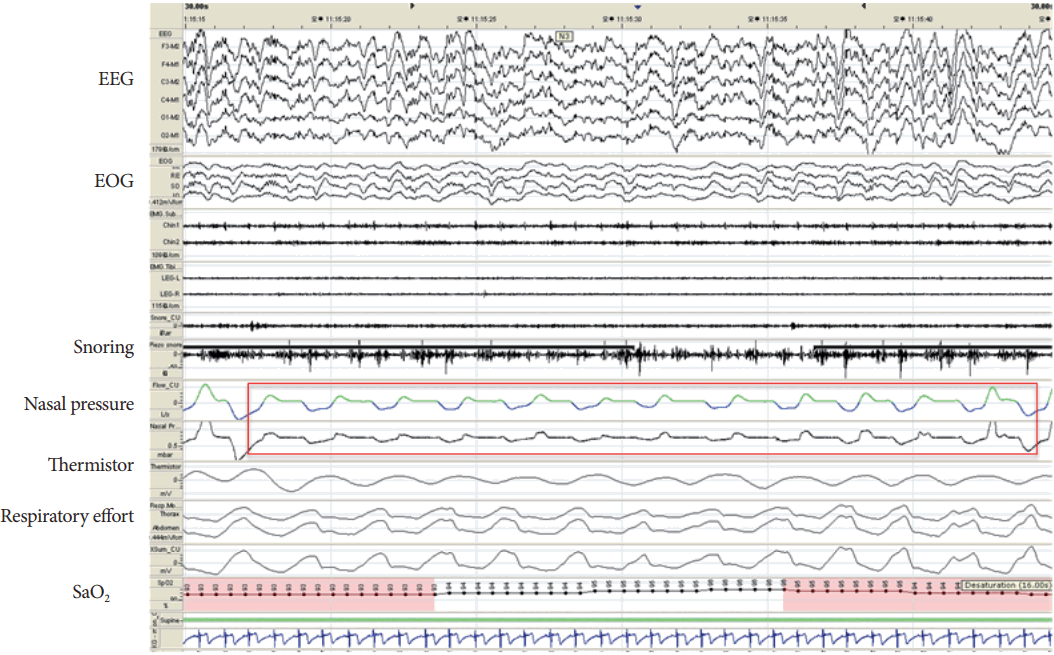
Night polysomnography (PSG) showed moderate to severe OSA (respiratory disturbance index 26.9/h, apnea-hypopnea index 22.9/h) with heavy snoring, tachypnea and bruxism (Table 1, Fig. 1, 2). Frequent hypopneas were observed during both light and deep NREM sleep (Fig. 1). The patient also had tachypnea with the respiratory rate of 26/ min (Fig. 2). Respiratory events were significantly more frequent in supine position. Obvious respiratory events were significantly decreased in lateral position but tachypnea and prolonged flow limitation still existed. Neither SW nor other behavior was observed during the PSG. No REM sleep without atonia was observed. Surgical treatment for tonsillar hypertrophy was favored to nasal continuous positive airway pressure (CPAP) therapy, taken into the patient’s irregular sleep schedule, the possibility of low adherence or poor compliance to CPAP, and his preference to surgical therapy. After tonsillectomy, snoring and apnea subsided and no more episode of SW was reported since then until he finished residency training despite frequent nights with insufficient sleep. Follow-up PSG was not performed. The recent follow-up phone call with the patient, 3 years from the surgery, confirmed no recurrence of SW.
Discussion
The pathophysiology of sleepwalking and other NREM parasomnia seems related to combination of two pathological processes leading to sleep-wake transition dysfunction, especially in adults [5]. The first is the phenomenon that deepens sleep and enhances sleep inertia, such as sleep deprivation and following slow wave sleep rebound [5,6]. The other is the condition that causes repeated cortical arousals or sleep fragmentation [5,6]. Impaired arousal mechanisms and the persistence of sleep drive may result in a failure of the brain to fully transit into wakefulness [6].
Those were described as a priming factor and a precipitating factor, respectively [6].
Our case is a clear example showing sleepwalking trigged by the combination of two factors in the patient whose sleepwalking remitted during late adolescence period.
Recent meta-analysis suggested relatively few people start sleepwalking in adulthood from the finding that the lifetime prevalence of SW does not significantly vary between childhood and adulthood [4].
The mechanism is not clear why preschool children have more common NREM parasomnia than adults. There is a theoretical assumption that synaptic pruning during childhood to remove redundant excessive synapses including GABAergic descending inhibition might contribute to activation of spinal cord central pattern generator [7].
Obstructive sleep apnea is known as a risk factor of NREM parasomnia in both children and adults [3,8,9]. The epidemiologic study for general population in the US using phone survey showed 3.6% of nocturnal wandering in adults. Individuals with OSA were at higher risk of frequent symptoms (odds ratio 3.9)[3]. There was evidence that successful treatment of OSA either with surgery or nasal CPAP in adult chronic sleepwalkers resolved sleepwalking [8]. Guilleminault et al. reported that 58% of 89 children who underwent night PSG showed additional diagnosis of sleep-disordered breathing (SDB) [9]. No more parasomnia was reported in those children who were surgically treated for SDB whereas untreated patients with SDB continued to have parasomnia [9]. Although NREM parasomnia is very common in children and considered as the benign phenomenon, night PSG should be considered for children with frequent parasomnia.
However, severe OSA frequently decreases slow wave sleep, especially in adults. It might not be sufficient to induce NREM parasomnia with OSA only. The case patient also did not develop parasomnia after spontaneous remission of sleepwalking until intern and residency training when he was prone to significant sleep deprivation. Rebound slow wave sleep following severe sleep deprivation may play an important role as a prerequisite condition to induce sleepwalking and complex nocturnal behaviors [10]. The experimental attempts to induce sleepwalking with forced arousals using auditory stimuli supported the priming effect of sleep deprivation [10]. In this study, no sleepwalking was induced in controls without sleepwalking history, suggesting the importance of genetic predisposition for NREM parasomnia [10].
Other triggers or associated condition with sleepwalking in adults include circadian rhythm sleep disorder, insomnia, alcohol abuse, psychiatric disorders and medication such as selective serotonin reuptake inhibitor, zolpidem, and sodium oxybate [3,7].
Adult sleepwalking can be potentially harmful, especially in medical personnel who are vulnerable to sleep deprivation. Eliminating triggers such as treating sleep apnea would be very important in predisposed individuals, not to mention avoiding sleep deprivation. Night PSG should be performed for differential diagnosis as well as scrutinizing risk factors or triggers in adult sleepwalkers.